By Bradley Stockwell
One of my favorite stories in all of physics is the story of sunlight because it touches on such a wide range of concepts. I apologize for the length of this post, but I guarantee you’ll be enlightened on many terms you probably hear thrown around a lot, but not a lot of people understand. Also, we learned in my previous post some of the important uses of light, but didn’t address the most important use of all, life!
Sunlight’s story, along with almost everything in the universe (we’ll ignore something called dark matter for now), begins with a hydrogen atom. Hydrogen is the most elementary and abundant element in the universe, hence the reason it is element one on the periodic table. Also because it’s comprised of one positively charged particle called a proton, which makes up its nucleus, and one negatively charged particle called an electron, which orbits around that single-proton nucleus. Within the sun, or any star, there is a process called nuclear fusion which transforms hydrogen into all 92 elements found in nature. Every grain of matter that makes up our physical world is forged in the heart of stars and is released when they begin to die. Not all stars produce all 92 elements however, like our sun will never get hot enough to fuse enough atoms together to produce heavy metals like gold. When I say heavy, what I am referring to is the element’s mass. The more sub-atomic particles shoved into an element’s nucleus, the heavier it is. Stars of different sizes produce different elements, but all stars begin with fusing hydrogen into helium as our sun is currently doing.
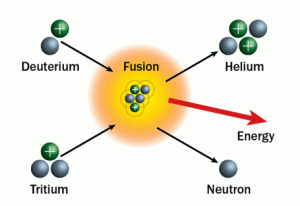
Within the sun’s core, hydrogen atoms are sped up from high amounts of energy, or heat, created by the force of the star’s mass on itself and collide at very high speeds, fusing them together to make helium. The sun has to smash four hydrogen atoms together to make one helium atom. The radioactive elements created in the steps in between are called hydrogen isotopes. Two hydrogen atoms make the stable isotope deuterium, three, the unstable isotope tritium, and four a helium atom. The difference between a stable and an unstable isotope is the even pairing of protons and neutrons (we’ll get to what a neutron is soon) in the nucleus. An even pairing, like one proton and one neutron (deuterium), is stable, but an uneven pairing, like one proton and two neutrons (tritium) is not and eventually falls apart into stable isotopes because it is too energetic to stay together. This ‘falling apart’ is known as radioactive decay.
Two hydrogen atoms make the stable isotope deuterium, three, the unstable isotope tritium, and four a helium atom.
If you’re a fan of The Simpsons, you may remember the Springfield baseball team was called The Isotopes. This was in reference to the town’s nuclear power plant, in which a forced and more violent version of this process occurs called nuclear fission. Typically uranium nuclei are loaded up with extra neutrons until it reaches what is called critical mass. Once critical mass is reached, the nuclei split and large amounts of energy are released because they can’t hold this new influx of neutrons. It’s kind of like your friend who drinks too much at a bar then spews all over the place. This neutron ‘spewing’ is what provides us with electrical power. While the process releases energy, it also leaves varying forms of unstable uranium isotopes that decay naturally into stable isotopes over sometimes hundreds of years. This is because uranium is such a heavier element in comparison to hydrogen, which its isotopes decay rather quickly. These radioactive leftovers are still highly energetic and emit damaging gamma and x-ray waves (we learned what these were in my previous post) and that is why containment is so crucial. My apologies for this nuclear fission tangent, but one should know the difference between fusion and fission. Fusion brings atoms together, fission rips them apart.
So back to the story of sunlight. When the sun does finally manage to smash four hydrogen atoms together, two of the hydrogen’s protons lose mass in the process and become neutrally charged particles called neutrons, making a total of two protons and two neutrons in the new helium nucleus with two orbiting electrons, one for each proton. The expelled proton mass, which eventually will become our beloved sunlight, is given off as energy in the form of highly energetic electromagnetic radiation (a.k.a. light) known as gamma rays. This is an excellent example of Einstein’s famous equation for energy, E=mc2, at work. What this equation says is mass (m) can be converted to energy (E). If you’ve ever tried to lose weight, the same concept applies. You’re trying to convert your mass into energy to lose it. However things on a quantum level work in funny ways. The neutron instead of being less massive actually becomes more massive than it was when it was a proton. This can be blamed on particles within protons and neutrons called quarks and how they behave; something I’ll leave for another post. The ‘C’ part of the equation stands for the speed of light constant which is just something that needs to be added formulaically in order to receive a correct calculation and we’ll get to why later.
We learned in my previous post that electromagnetic radiation is made of particles called photons. These newly created gamma ray photons are at first far too dangerous for earthly consumption. However after tens of thousands of years of being passed around between densely packed atoms within the sun, the photons tire out a bit until they become less energetic visible light photons, or what we call sunshine. Even traveling at the speed of light, photons can take up to a million years to escape the sun; a distance of 432,000 miles from core to surface. While this may seem like a long distance, compare it to the 93 million miles photons travel in only 8 minutes and it becomes apparent how abated those photons are by being continually absorbed and emitted by the soup of atoms within the sun. However once they hit the empty vacuum of space, they have a straight shot to Earth.
When photons finally enter Earth’s atmosphere, some of them are absorbed by tiny pores on plants’ leaves called stomata that convert those photons into chemical energy. This is done by the synthesizing of hydrogen atoms from water in the plant with carbon dioxide in the air to create sugars. This process, I’m sure you’re familiar with, is called photosynthesis. Since plants only use the hydrogen from water, they emit the remaining oxygen as a waste product and we literally breathe their shit. The sugar is stored and later converted into kinetic energy to allow the plant to function. This sugar however can be transferred to a creature that eats the plant and a creature that eats that creature and so forth. Animals (including us) extract energy from these sugars by reacting them with the oxygen they breathe and exhale the remaining carbon dioxide from the sugars so that another plant can use it to create more sugar and oxygen for them to consume.
So next time you look up at the sun (not directly!), think about what’s going on inside there. Think about everything nuclear fusion gives you; air, food— the very matter you’re made of, and say thanks. And as the sunlight warms your skin, think about the tens of thousands of years it took for those photons to reach it. And here’s another interesting fact to blow your mind on; for those photons, you are their entire existence! Well at least within our idea of existence. This is where the ‘C’ (the speed of light constant) in E=mc2 comes into play. The photon, which is energy, travels at the speed of light and that is why that speed needs to be figured into every calculation for energy. It is ‘constant’. And according to Einstein’s theory of relativity, time slows down the faster you move relative to another object until it completely stops at the speed of light. The photon’s time, relative to ours, doesn’t exist. The photon is considered timeless . . . well at least until it’s brought into our reality when you absorbed it as heat. I’ll segue this into my next post which will be on the theory of relativity and quantum mechanics. Until then, stay curious my friends!




Leave a Reply